Stereochemistry Practice Problems
This comprehensive set of practice problems on Stereochemistry includes questions on determiningR andS configuration,R andS configuration in Fischer and Newman projections, and determining the relationship between molecules such as enantiomers, diastereomers, and constitutional isomers. There are also questions on optical activity, such as determining the optical rotation, the enantiomeric excess, the percentage of each enantiomer, and more. Towards the end, we also practice meso compounds, Fischer projections, converting between Fischer and Newman projections and vice versa.
This is quite a lot, but we have even more. As a CS member, you will also get access to this over 100-question multiple-choice stereochemistry quiz and all the quizzes as well.
This video is a fragment thequiz and explains therules for drawing Fischer projections in the context of determining the relationship between given compounds. The quiz comes with a 3-hour video solution.
Practice
Identify all the chiral centers in each molecule and determine the absolute configuration asR orS:

This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
Identify all the chiral centers in eachFischer projection and determine the absolute configuration asR orS:

This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
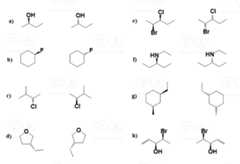
For each of the following pairs of compounds, determine the relationship between the two compounds: Are theyenantiomers or the same compound drawn differently? If you hesitate, determine theabsolute configuration of chiral centers (if any: R orS).
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
PracticingR and S is never too much. This1.5-hour video is a collection of examples taken from themultiple choice quizzes determining theR andS configuration in the context of naming compounds, determining the relationship between compounds, and chemical reactions.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
Determine whether each pair of compounds areEnantiomers orDiastereomers: Assigning the absolute configuration (R or S) will be very helpful.

This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
Draw a diastereomer for each of the following compounds:


Determine the relationship in each of the following pairs. Do the drawings representconstitutional isomersor stereoisomers, or are they just different ways of drawing the same compound? If they are stereoisomers, are theyenantiomersor diastereomers?
Explain your answer by converting the drawings into the same representation, i.e. if you are comparing aNewman projection to aFischer projection, you need to convert both into either a Newman or Fischer projection. Assign all theabsolute configurations asR orS if you hesitate.

This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
What is the relationship in each of the following pairs of compounds. Are they identical,constitutional isomers, enantiomers , diastereomers or there is none of these?

This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
1,4-Disubstituted Cyclohexanes
What is the relationship in each of the following pairs:

a, b – both are pairs of diastereomers. Find the plane of symmetry for each molecule to confirm they are achiral diastereomers and then check them in the solution.
Unlike, these two, c represents two chiral molecules which are enantiomers.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
Practice problems are never too much! Here is another set of enantiomers, diastereomers, and constitutional isomers:
What is the relationship between each of the following pairs of molecules? Indicate whether they are identical, constitutional isomers, enantiomers, diastereomers, or none of these.

The answers can be foundhere.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
What is the enantiomeric excess of an adrenaline sample that has a specific rotation of -47.7? Pure adrenaline has a specific rotation of -53.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
How many percent of cholesterol and its enantiomer are present in a sample with an observed specific rotation of -22.4°? The specific rotation of pure cholesterol is -32°.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
The specific rotation of a pure substance is +78°. What is the specific rotation of a mixture containing 75% of this isomer and 25% of the (-) isomer?
a) +78°
b) +39°
c) +59°
d) +20°
e) 0°
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
(+)-Cavicularin has a specific rotation of +168.2°. What would be the specific rotation of a solution which contains 60.0% (-)-Cavicularin and 40.0% (+)-Cavicularin?
a) +33.6°
b) +101°
c) -33.6°
d) -101°
e) -67.3°
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
The specific rotation of (S)-alanine is +8.5°. If a mixture of alanine enantiomers is 80%S and 20%R, what is the specific rotation of the mixture?
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
All the molecules shown below are achiral because of the presence of a plane of symmetry. Confirm that they are achiral by drawing the corresponding plane of symmetry and determine if they are meso compounds. Remember that the plane of symmetry can slice atoms in half.

This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
Determine whether each of the molecules is chiral or achiral. Explain your answer by showing the appropriate symmetry element:

This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
Fischer Projections
Convert the followingbond-line structure to the corresponding Fischer projection.


This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
Somemore practice example of the Fischer projection
Convert the following Bond-Line, Newman and Fischer projections as indicated below:
The answers can be found under the following article:
Converting Bond-Line, Newman Projection, and Fischer Projections
The following questions are from theStereochemistry Quiz and the video is a fragment of a 3-hour solution.
Select the enantiomer for the following compound:
a)

b)

c)

This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
Which of the following is a meso compound?

This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
What is the relationship between these two structures?
a)

b)

This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to theanswers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps,and the powerful set ofOrganic Chemistry 1 and 2 Summary Study Guides.
Check Also
- How to Determine the R and S Configuration
- The R and S Configuration Practice Problems
- What is Nonsuperimposable in Organic Chemistry
- Chirality and Enantiomers
- Diastereomers-Introduction and Practice Problems
- Cis and Trans Stereoisomerism in Alkenes
- E and Z Alkene Configuration with Practice Problems
- Enantiomers vs Diastereomers
- Enantiomers Diastereomers the Same or Constitutional Isomers with Practice Problems
- Configurational Isomers
- Optical Activity
- Specific Rotation
- Racemic Mixtures
- Enantiomeric Excess (ee): Percentage of Enantiomers from Specific Rotation with Practice Problems
- Symmetry and Chirality. Meso Compounds
- Fischer Projections with Practice Problems
- R and S Configuration in the Fischer Projection
- R and S configuration on Newman projections
- R and S Configuration of Allenes
- Converting Bond-Line, Newman Projection, and Fischer Projections
- Resolution of Enantiomers: Separate Enantiomers by Converting to Diastereomers
- Stereochemistry Practice Problems Quiz
4 thoughts on “Stereochemistry Practice Problems”
Hi, in question 8- b why Br is pointing towards but in f Cl and F are pointing away? both b and f they have a identical bond?
and second question if we want to flip a molcule in a bond line dash becomes a wedge? or always remain dash a dash after fliping?
Thank you in advance
ReplyHi there,
For the first question, I’ve already responded to it under another post, but in case you missed it, I’m quoting my answer here again. Please don’t hesitate to let me know if you’d like further clarification.
“I am assuming you are referring to the fact that “towards us” inbond-line notation indicates a wedge, but here it is a dash. The difference in bond-line andFischer notations is the view angle.In Fischer, we are looking from the top, andboth wedge and dash are pointing towards us, whereas in bond-line, we are looking from the side, and it is the wedge group that is pointing towards us.
See how the wedge Br and dash H are pointing towards us when converting to a Fischer projection in this example:
Check alsothis article for more examples on converting Bond-line to Fischer projection and vice versa.”
Question 2: Whether you need to flip the wedges and dashes depends on what you are trying to represent and from which angle you are viewing the molecule. There is no single rule that always requires changing or keeping them.
•Same molecule, different orientation: if you rotate the molecule 180° in the plane of the paper, the wedges and dashes stay as they are, but the atom/groups that were pointing up will now point down, and vice versa.
•If you flip the molecule outside of the paper, then yes, all the wedges and dashes will be switched.
•The Enantiomer: if you want to draw the mirror image, keep the zigzag backbone the same and flip all the wedges and dashes. See this image from the post “Enantiomers vs Diastereomers.”:
I recommend using a molecular model and physically rotating it to see how the bond projections change. Wedge and dash notation is relative because it depends on your viewing angle. What ultimately matters is the absolute configuration (R/S) at each chiral center. It may not be practical to determine the R and S every single time, and there is still a chance of making a mistake in doing that, so having a model to improve 3D visualization would be very beneficial.
Reply





