Pancreatic Cancer: A Review
- PMID:34547082
- PMCID: PMC9363152
- DOI: 10.1001/jama.2021.13027
Pancreatic Cancer: A Review
Erratum in
- Error in Box.[No authors listed][No authors listed]JAMA. 2021 Nov 23;326(20):2081. doi: 10.1001/jama.2021.19984.JAMA. 2021.PMID:34812887Free PMC article.No abstract available.
Abstract
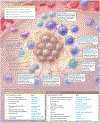
Importance: Pancreatic ductal adenocarcinoma (PDAC) is a relatively uncommon cancer, with approximately 60 430 new diagnoses expected in 2021 in the US. The incidence of PDAC is increasing by 0.5% to 1.0% per year, and it is projected to become the second-leading cause of cancer-related mortality by 2030.
Observations: Effective screening is not available for PDAC, and most patients present with locally advanced (30%-35%) or metastatic (50%-55%) disease at diagnosis. A multidisciplinary management approach is recommended. Localized pancreas cancer includes resectable, borderline resectable (localized and involving major vascular structures), and locally advanced (unresectable) disease based on the degree of arterial and venous involvement by tumor, typically of the superior mesenteric vessels. For patients with resectable disease at presentation (10%-15%), surgery followed by adjuvant chemotherapy with FOLFIRINOX (fluorouracil, irinotecan, leucovorin, oxaliplatin) represents a standard therapeutic approach with an anticipated median overall survival of 54.4 months, compared with 35 months for single-agent gemcitabine (stratified hazard ratio for death, 0.64 [95% CI, 0.48-0.86]; P = .003). Neoadjuvant systemic therapy with or without radiation followed by evaluation for surgery is an accepted treatment approach for resectable and borderline resectable disease. For patients with locally advanced and unresectable disease due to extensive vascular involvement, systemic therapy followed by radiation is an option for definitive locoregional disease control. For patients with advanced (locally advanced and metastatic) PDAC, multiagent chemotherapy regimens, including FOLFIRINOX, gemcitabine/nab-paclitaxel, and nanoliposomal irinotecan/fluorouracil, all have a survival benefit of 2 to 6 months compared with a single-agent gemcitabine. For the 5% to 7% of patients with a BRCA pathogenic germline variant and metastatic PDAC, olaparib, a poly (adenosine diphosphate [ADB]-ribose) polymerase inhibitor, is a maintenance option that improves progression-free survival following initial platinum-based therapy.
Conclusions and relevance: Approximately 60 000 new cases of PDAC are diagnosed per year, and approximately 50% of patients have advanced disease at diagnosis. The incidence of PDAC is increasing. Currently available cytotoxic therapies for advanced disease are modestly effective. For all patients, multidisciplinary management, comprehensive germline testing, and integrated supportive care are recommended.
Figures

Comment in
- A Review of Pancreatic Cancer.Neoptolemos JP, Springfeld C, Hackert T.Neoptolemos JP, et al.JAMA. 2021 Dec 21;326(23):2436. doi: 10.1001/jama.2021.20065.JAMA. 2021.PMID:34932083No abstract available.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
